
The reducing ability of the metals grows weaker while traversing down the series. These metals tarnish/corrode very easily. (b) What would you observe when you put (i) some zinc pieces into blue copper sulphate solution (ii) some copper pieces into green ferrous sulphate solution (c ) Name a metal which combines with hydrogen gas. The metals at the top of the reactivity series are powerful reducing agents since they are easily oxidized. (a) What is the activity series of metals Arrange the metals Zn, Mg, Al, Cu and Fe in a decreasing order of reactivity. Long Tabular Form of the Reactivity Series Reactivity Series of Metals Ions Formed Lead Pb2+ Hydrogen H+ (Non-Metal, Reference for Comparison) Antimony Sb3+ Bismuth Bi3+ What is the effect of reactivity series on reducing power? What is the tabular form of the reactivity series?  
Therefore, the reactivity series of metals can be used to predict the reactions between metals and water. How can the reactivity series be used to predict chemical reactions?įor example, the reaction between potassium and water yields potassium hydroxide and H 2 gas, as described by the chemical equation provided below. The data provided by the reactivity series can be used to predict whether a metal can displace another in a single displacement reaction. The reactivity series of metals, also known as the activity series, refers to the arrangement of metals in the descending order of their reactivities. It states like : Katrina ne car mangi Alto, zen, ferrari phirbhi kyu milli silver Audi. Most suitable and easy way to remember reactivity series (Hindi). 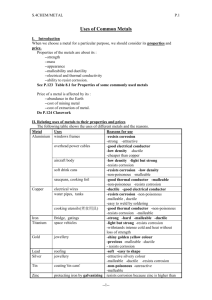
How do you remember the spectrochemical series? If you carry on with physics at uni you’ll find that you learn how to explain/understand quite a few things that just have to be limited to memorization in A-level chemistry. There are several reactivity series mnemonics to help you remember the order of the metals One that we like goes as follows: Please send lions, cats, monkeys. Yes it is mostly memorization at A level. Organic Chemistry: It shouldn’t surprise you that organic chemistry takes the No. Which is the correct order of reactivity series Mg Ca Na K?Īnswer: Option (b) K < Na < Ca < Mg is correct answer. A metal placed at tire top of the activity series would displace metal below it. It can also be used to obtain information on the. A metal placed above hydrogen in the activity series will displace hydrogen from water or acids. The reactivity series of metals, also known as the activity series, refers to the arrangement of metals in the descending order of their reactivities. It shows you how to tell if a single replacement reaction will work or if there will be no. The arrangement of metals in the vertical column in the order of decreasing reactivity is called reactivity series or activity series. This chemistry video tutorial explains the activity series of metals and elements such as hydrogen. What is reactivity series How does the reactivity series? I’d learn it just to stay on the safe side, when I did GCSEs we had to learn the reactivity series so I don’t know if it’s changed for you. It says you need to “explain” things by using the reactivity series but it doesn’t say you have to “recall” it. I LIKE HIS CAR’S SILVERY GATE.ĭo you need to Memorise the reactivity series? In this article, the preparation, electronic properties and design of metal acyclic carbene complexes with different functional properties for the development of advanced materials are described. The environmental sensitivity of the properties of these complexes also made them ideal for the development of stimuli-responsive materials and chemical sensors. As a result, the functional properties of acyclic complexes can be drastically varied by rational molecular design of the ligands. Moreover, the open structure of acyclic carbene ligands has made their structure and the electronic properties strongly dependent on the substituents as well as the micro-environment. As transition-metal acyclic carbene complexes can be prepared from metal isocyanide synthetic precursors, substituents of different electronic and steric natures as well as functional moieties can be readily introduced into acyclic carbene ligands by changing the isocyanide ligand. Therefore, transition-metal acyclic carbene complexes are considered viable alternatives to NHC complexes in the development of metal-based functional materials. As acyclic carbene ligands show strong σ-donating properties comparable to N-heterocyclic carbene (NHC) ligands, transition-metal complexes with acyclic carbene ligands also demonstrate outstanding performance and functional properties similar to their NHC counterparts. Transition-metal acyclic carbene complexes have received increasing attention in recent years.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
